FDA OKs ‘life-changing’ gene therapy for hearing loss — and it’s free
By Mckenzie Beard
Published on April 24, 2026.
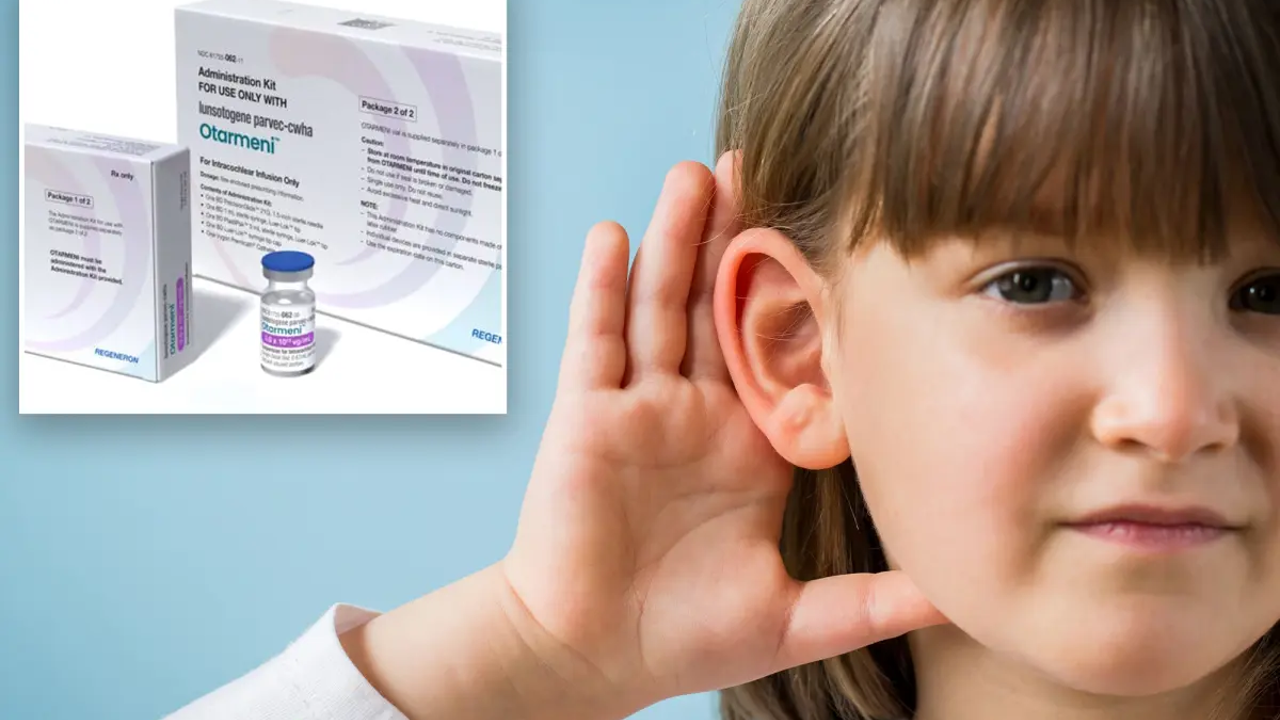
The US Food and Drug Administration has approved the first gene therapy for a rare form of hearing loss, Otarmeni, by Regeneron, the maker of the drug, that will be free for US children and adults. The drug is designed for people born deaf due to a mutation in a gene called OTOF, which normally helps the body make a protein called otoferlin, which is needed to hear. Without this, the hair cells in the inner ear can't send sound signals to the brain, leading to severe to total deafness from birth. The FDA fast-tracked the treatment through its National Priority Voucher program, which speeds up reviews for products targeting major unmet medical needs. While the treatment applies to only a small percentage of people with hearing loss and may not fully restore natural sound, experts believe it could potentially open the door to more gene therapies.


