FDA removes menopause hormone therapy warning after 20 years, opening new options for women
By Denise
Published on March 25, 2026.
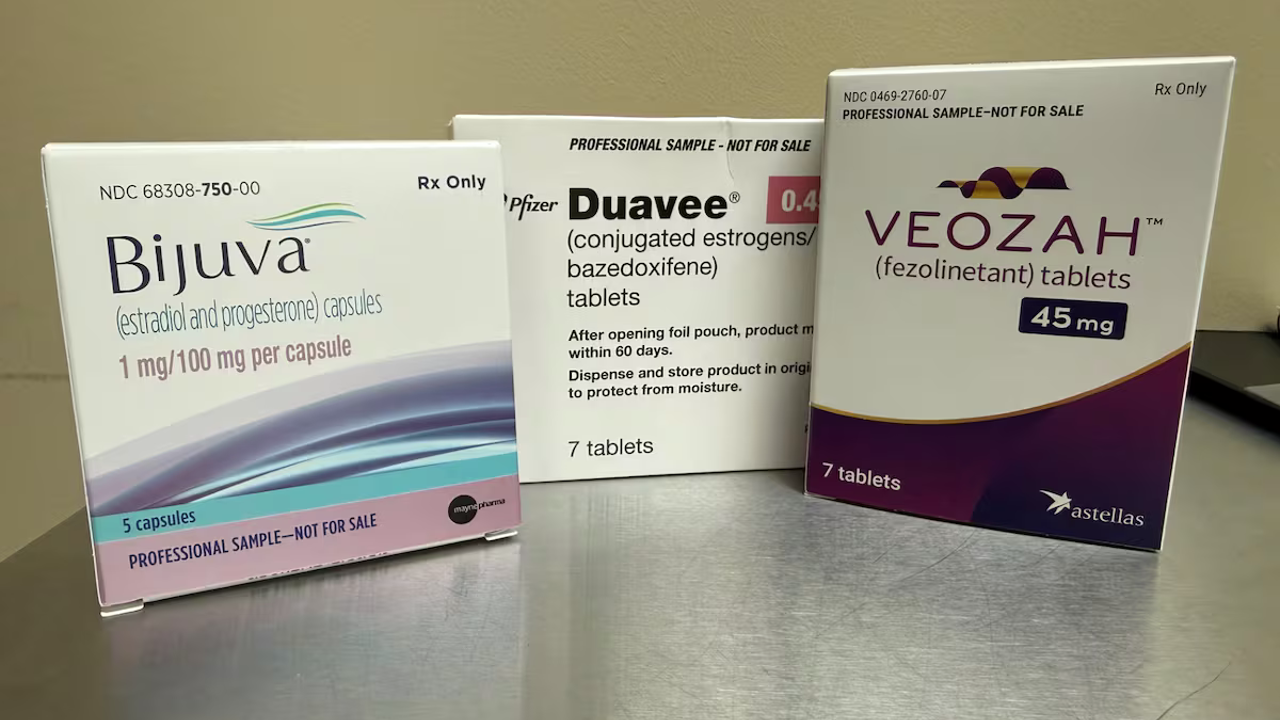
The FDA has removed labels from six hormone replacement products, potentially restoring treatment options for menopausal women who were previously discouraged from using hormone replacement therapy. The changes were approved by the American College of Obstetricians & Gynecologists. The FDA's black box warning alerts health care providers and patients about potentially serious or life-threatening risks. However, the FDA did not ask drug sponsors to remove risks of cardiovascular disease and breast cancer from the labels “Warnings and Precautions” nor did the boxed warning for endometrial cancer for systemic estrogen-alone products. The American Society of Health-System Pharmacists reported a shortage of estrogen patches. The labeling change eliminated a “blanket warning” and reduced the “angst” people might experience when taking hormone replacements, especially patients likely to benefit.


