Doctor explains FDA's refusal to endorse drug once touted as autism treatment
By Ed Harding
Published on March 10, 2026.
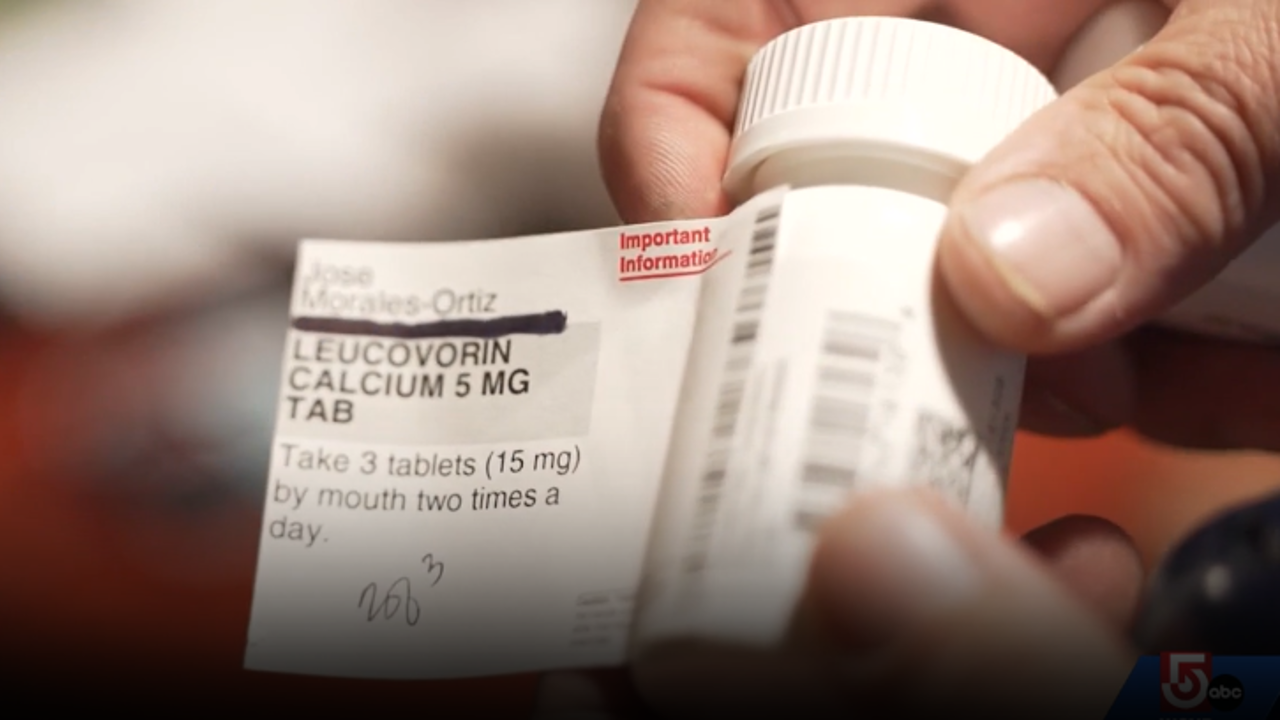
The FDA has declined to endorse the drug, LeucoVorin, once touted as a potential treatment for autism. The drug is currently being used for a rare form of CEREBRAL FOLATE DEFICIENCY, a rare disease that affects 1 in 1,000,000 people. However, the FDA has stated that there is not enough EVIDENCE to support this claim. Doctor, Todd ELLERIN, explains that while there was a significant SPIKE in LEUCOVORIN PREScriptTIONS, there is no evidence to support it for the large percentage of children with autism. He also notes that the drug's use in pre-natal time can lead to other health issues, including a decrease in the use of TYLENOL for pregnant women.