FDA-approved drug slows progression of dry macular degeneration
By Gina Glaros
Published on March 23, 2026.
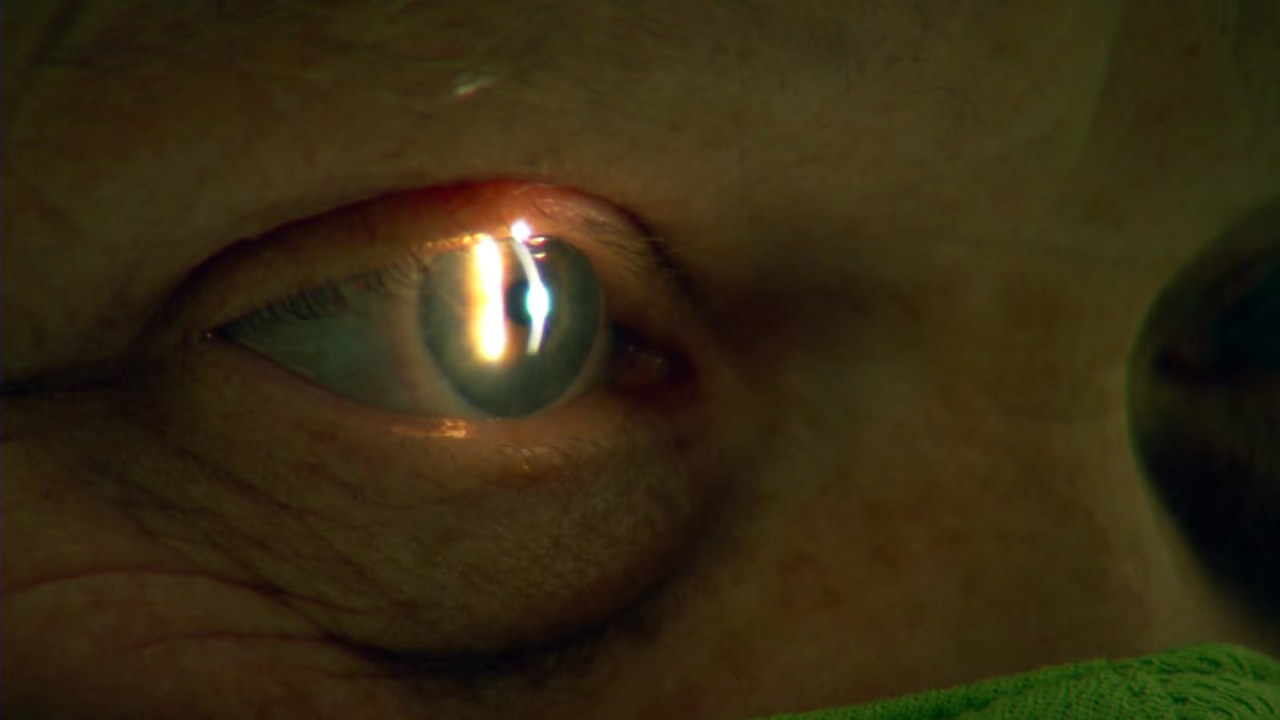
The most common type of macular degeneration, dry, occurs when small deposits form under the retina, causing cells in the eye to die off. There is currently no cure for this disease, but in 2023, the first medicine approved by the U.S. Food and Drug Administration is expected to slow the progression of this disease. Dr. Christopher D. Riemann, a vitreoretinal surgeon at the Cincinnati Eye Institute, is part of a clinical trial testing whether injecting human retinal pigment epithelium cells into the eyes of patients can improve vision. Researchers are also exploring an innovative electronic eye implant paired with augmented-reality glasses.


