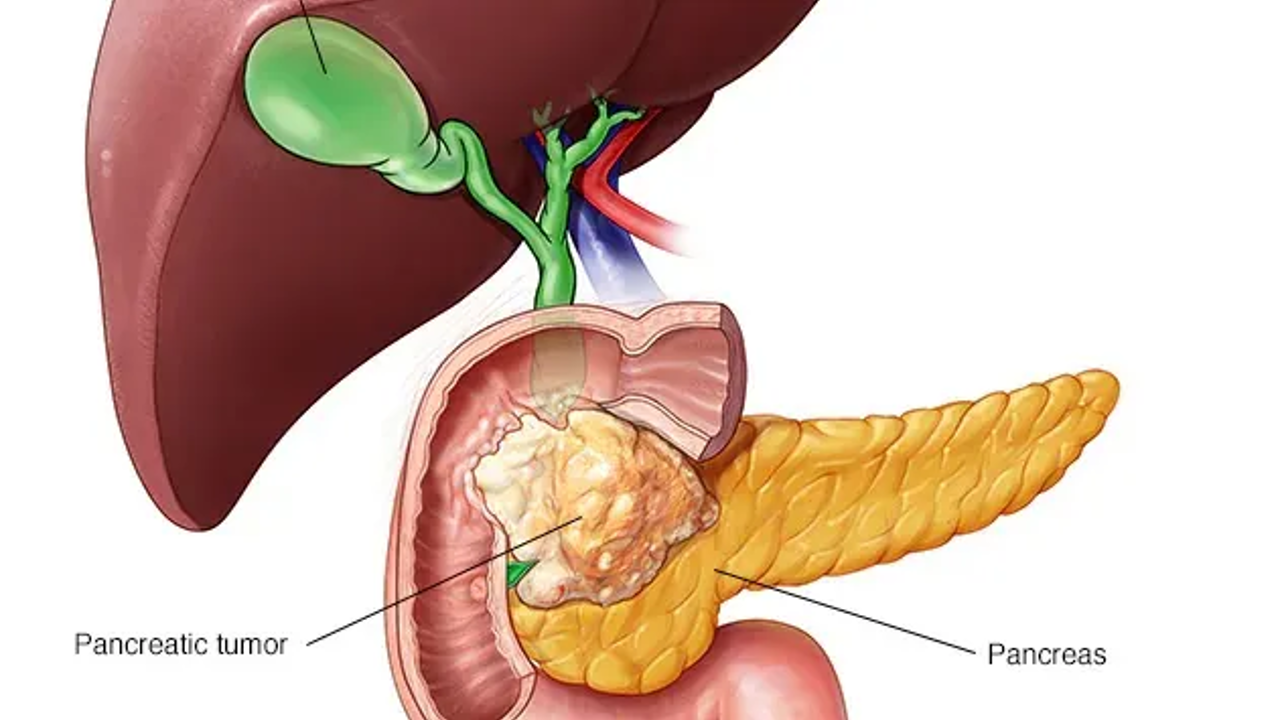
FDA green lights early access to pancreatic cancer drug, daraxonrasib
By Ken Alltucker
Published on May 1, 2026.
The Food and Drug Administration (FDA) has approved the experimental pancreatic cancer drug daraxonrasib, which has not yet been approved by the FDA. The company had released promising late-stage clinical trial results in April for patients with pancreatic cancers that had spread to other parts of the body. The FDA has allowed Revolution Medicines to begin an "expanded access treatment protocol" for some patients who had previously been treated for pancreatic disease. Former Sen. Ben Sasse, R-Nebraska, called darxonrasIB a "miracle drug" for helping him manage his pancreatic health. The National Cancer Institute estimates that more than 52,000 people will die from pancreatan cancer in 2026.


